Is Carbon Dioxide a Polar Molecule? A Comprehensive Guide to Understanding CO2 Polarity
Carbon dioxide (CO2) is one of the most studied molecules in chemistry, and its polarity is a common topic of discussion among students and researchers alike. Understanding whether carbon dioxide is polar or nonpolar requires a deep dive into its molecular structure, bond characteristics, and dipole moments. This article will explore the properties of CO2 and provide a clear answer to the question: Is carbon dioxide a polar molecule?
In this article, we will explore the molecular structure of carbon dioxide, its chemical bonds, and its dipole moments. By examining these factors, we can determine whether CO2 is polar or nonpolar and understand the implications of this classification in various scientific and environmental contexts.
Carbon dioxide plays a critical role in our atmosphere and global climate. Its behavior as a molecule affects everything from atmospheric chemistry to industrial applications. Let’s delve into the science behind CO2 polarity and uncover the truth about its classification.
Molecular Structure of Carbon Dioxide
The molecular structure of carbon dioxide is linear, with one carbon atom bonded to two oxygen atoms. This arrangement is crucial in determining the polarity of the molecule. In a linear structure, the atoms are positioned in a straight line, which influences how the molecule interacts with electric fields.
Key Features of CO2 Structure:
- Linear geometry
- Two double bonds between carbon and oxygen
- Symmetrical arrangement of atoms
This symmetry plays a significant role in the overall polarity of the molecule. The linear structure ensures that any dipole moments created by the bonds cancel each other out, as we will explore in more detail later.
Chemical Bonding in CO2
Types of Bonds in Carbon Dioxide
Carbon dioxide consists of covalent bonds between carbon and oxygen atoms. Specifically, there are two double bonds in the molecule. These double bonds are formed by the sharing of four electrons between the carbon and oxygen atoms.
Characteristics of CO2 Bonds:
- Strong covalent bonds
- Electronegativity difference between carbon and oxygen
- Bond length of approximately 1.16 Å
The electronegativity difference between carbon (2.55) and oxygen (3.44) creates a polar bond. However, the overall polarity of the molecule depends on the arrangement of these bonds and the resulting dipole moments.
Dipole Moment of Carbon Dioxide
Understanding Dipole Moments
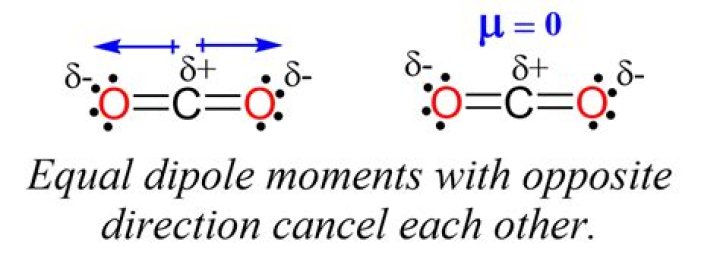
A dipole moment is a measure of the separation of positive and negative charges within a molecule. In carbon dioxide, each C=O bond has a dipole moment due to the difference in electronegativity between carbon and oxygen. However, because the molecule is linear and symmetrical, the dipole moments cancel each other out.
Why Do Dipole Moments Cancel in CO2?
- Symmetrical arrangement of bonds
- Equal and opposite dipole directions
- Linear geometry ensures no net dipole
As a result, the overall dipole moment of carbon dioxide is zero, making it a nonpolar molecule despite having polar bonds.
Is Carbon Dioxide Polar or Nonpolar?
Based on the molecular structure, chemical bonding, and dipole moments, carbon dioxide is classified as a nonpolar molecule. Although the individual C=O bonds are polar, the symmetrical arrangement of the molecule ensures that the dipole moments cancel out, resulting in no net polarity.
Factors Influencing CO2 Polarity:
- Molecular geometry
- Dipole moment cancellation
- Symmetrical distribution of charges
This classification is important in understanding how carbon dioxide interacts with other molecules and its behavior in various environments.
Environmental Implications of CO2 Polarity
CO2 and Climate Change
The nonpolar nature of carbon dioxide does not directly affect its role in climate change. However, understanding its molecular properties helps scientists predict how CO2 behaves in the atmosphere. As a greenhouse gas, carbon dioxide traps heat and contributes to global warming, regardless of its polarity.
Key Environmental Facts About CO2:
- Contributes significantly to the greenhouse effect
- Increases in concentration due to human activities
- Impacts global temperature and weather patterns
While the polarity of CO2 may not be a primary factor in climate science, its molecular structure and behavior are essential in understanding its environmental impact.
Applications of CO2 in Industry
Uses of Carbon Dioxide
Carbon dioxide has numerous industrial applications, ranging from food and beverage production to fire extinguishers. Its nonpolar nature makes it suitable for various processes where polarity is not a limiting factor.
Common Applications of CO2:
- Carbonation in beverages
- Fire suppression systems
- Supercritical CO2 extraction
These applications highlight the versatility of carbon dioxide and its importance in modern industries.
Common Myths About CO2 Polarity
There are several misconceptions about the polarity of carbon dioxide. Some people assume that because CO2 has polar bonds, it must be a polar molecule. However, as we have seen, the symmetrical structure of the molecule cancels out the dipole moments, making it nonpolar.
Common Myths:
- CO2 is polar because it has polar bonds
- Polarity affects its role in climate change
- Nonpolar molecules cannot interact with water
Dispelling these myths is crucial for a deeper understanding of carbon dioxide and its properties.
The Science Behind Molecular Polarity
Factors Influencing Polarity
Molecular polarity depends on several factors, including molecular geometry, electronegativity differences, and dipole moments. By analyzing these factors, scientists can classify molecules as polar or nonpolar.
Key Factors in Determining Polarity:
- Molecular shape and symmetry
- Electronegativity differences between atoms
- Dipole moment calculations
Understanding these principles helps in predicting the behavior of molecules in various environments and applications.
Comparing CO2 with Other Polar Molecules
Water vs. Carbon Dioxide
While carbon dioxide is nonpolar, water (H2O) is a classic example of a polar molecule. The difference lies in their molecular structures and dipole moments. Water has a bent geometry, which prevents dipole moments from canceling out, whereas CO2’s linear structure ensures complete cancellation.
Comparison of CO2 and H2O:
- CO2: Linear, nonpolar
- H2O: Bent, polar
- Both have polar bonds, but differ in overall polarity
This comparison highlights the importance of molecular geometry in determining polarity.
Conclusion: Understanding Carbon Dioxide Polarity
In conclusion, carbon dioxide is a nonpolar molecule due to its symmetrical linear structure and the cancellation of dipole moments. Despite having polar bonds, the overall polarity of the molecule is zero, making it nonpolar. Understanding the science behind molecular polarity is essential in various fields, from chemistry to environmental science.
We invite you to share your thoughts and questions in the comments section below. If you found this article helpful, please consider sharing it with others. For more insights into chemistry and related topics, explore our other articles on the site.
References:
- Atkins, P. W., & de Paula, J. (2018). Atkins' Physical Chemistry. Oxford University Press.
- Brown, T. L., LeMay, H. E., Bursten, B. E., & Murphy, C. J. (2018). Chemistry: The Central Science. Pearson.
- IPCC. (2021). Climate Change 2021: The Physical Science Basis. Cambridge University Press.



