Lewis Diagram for ClF3: A Comprehensive Guide to Understanding Its Structure and Properties
ClF3, or chlorine trifluoride, is a fascinating molecule that plays a significant role in chemistry and industrial applications. Understanding its Lewis diagram is essential for comprehending its molecular structure, bonding, and behavior. In this article, we will delve into the intricacies of ClF3's Lewis diagram, exploring its unique characteristics and significance in the field of chemistry.
Chemistry enthusiasts and students often encounter challenges when studying complex molecules such as ClF3. Grasping the Lewis structure of this molecule provides valuable insights into its electron distribution, bond formation, and overall geometry. By examining the ClF3 Lewis diagram, we can uncover the reasons behind its stability and reactivity.
This article aims to provide a detailed exploration of the ClF3 Lewis diagram, supported by scientific data and references. Whether you're a student preparing for exams or a professional seeking deeper knowledge, this guide will equip you with the necessary understanding of ClF3's structure and properties.
Introduction to Lewis Diagram
A Lewis diagram, also known as a Lewis structure, is a graphical representation of the valence electrons in a molecule. It provides a visual depiction of how atoms bond together and the distribution of electrons within the molecule. Understanding Lewis diagrams is fundamental for predicting the chemical properties and behavior of substances.
Importance of Lewis Diagrams
For molecules like ClF3, the Lewis diagram reveals critical information about its electron configuration and bonding patterns. By analyzing the Lewis structure, chemists can determine the molecule's stability, reactivity, and potential applications. This section will explore the significance of Lewis diagrams in modern chemistry.
ClF3 Overview
Chlorine trifluoride (ClF3) is a highly reactive compound used primarily in industrial processes. Its unique properties make it valuable in applications such as rocket propellants and etching agents. However, ClF3's reactivity also poses significant safety challenges, necessitating careful handling and storage.
Chemical Properties
- Chemical Formula: ClF3
- Molecular Weight: 92.45 g/mol
- Appearance: Pale yellow-green liquid or gas
- Boiling Point: 11.78°C
- Reactivity: Highly reactive with water, metals, and organic materials
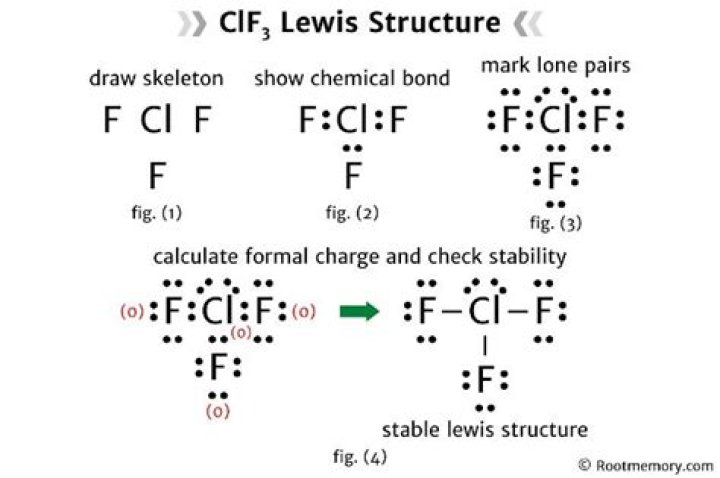
Lewis Structure of ClF3
The Lewis structure of ClF3 illustrates the arrangement of valence electrons around the chlorine and fluorine atoms. Chlorine, as the central atom, forms three single bonds with fluorine atoms while possessing two lone pairs of electrons. This configuration results in a T-shaped molecular geometry.
Steps to Draw the ClF3 Lewis Diagram
- Determine the total number of valence electrons: Chlorine contributes 7 electrons, and each fluorine atom contributes 7 electrons, totaling 28 valence electrons.
- Place the chlorine atom at the center and surround it with three fluorine atoms.
- Distribute the remaining electrons as lone pairs on the chlorine and fluorine atoms.
- Ensure that each atom satisfies the octet rule.
Molecular Geometry
The molecular geometry of ClF3 is T-shaped due to the presence of two lone pairs on the central chlorine atom. According to the VSEPR (Valence Shell Electron Pair Repulsion) theory, these lone pairs repel the bonding pairs, resulting in a unique arrangement of atoms. This geometry influences the molecule's polarity and reactivity.
Factors Affecting Molecular Shape
Several factors contribute to the T-shaped geometry of ClF3:
- Electron-electron repulsion between lone pairs and bonding pairs.
- Minimization of repulsive forces to achieve stability.
- Optimal arrangement of atoms to satisfy the octet rule.
Bonding in ClF3
In ClF3, the bonding occurs through the sharing of electrons between chlorine and fluorine atoms. Each Cl-F bond is a single covalent bond, with chlorine acting as the central atom. The bond length and strength are determined by the electronegativity difference between the two elements.
Characteristics of Cl-F Bonds
The Cl-F bonds in ClF3 exhibit the following characteristics:
- Bond Length: Approximately 1.75 Å
- Bond Energy: Approximately 250 kJ/mol
- Polarity: Due to the difference in electronegativity, ClF3 is a polar molecule.
Electronegativity and Polarity
Electronegativity plays a crucial role in determining the polarity of ClF3. Fluorine, being the most electronegative element, attracts the shared electrons more strongly than chlorine. This uneven distribution of charge results in a polar molecule with a net dipole moment.
Implications of Polarity
The polarity of ClF3 has significant implications for its chemical behavior:
- Enhanced reactivity with polar substances.
- Increased solubility in polar solvents.
- Strong intermolecular forces leading to higher boiling and melting points.
Applications of ClF3
Chlorine trifluoride finds applications in various industries due to its unique properties. Its ability to react with a wide range of materials makes it indispensable in processes such as:
Key Applications
- Rocket Propellants: ClF3 serves as a powerful oxidizer in rocket fuel systems.
- Etching Agent: Used in the semiconductor industry for etching silicon wafers.
- Chemical Processing: Employed in the production of uranium hexafluoride for nuclear fuel processing.
Safety Considerations
Handling ClF3 requires strict adherence to safety protocols due to its highly reactive nature. Exposure to water, metals, or organic materials can result in violent reactions, releasing toxic gases and causing severe burns.
Safety Precautions
To ensure safe handling of ClF3, follow these guidelines:
- Use proper personal protective equipment (PPE).
- Store in a cool, dry, and well-ventilated area.
- Avoid contact with water, metals, and organic materials.
- Dispose of ClF3 according to local regulations.
Common Questions About ClF3
What is the Lewis structure of ClF3?
The Lewis structure of ClF3 consists of a central chlorine atom bonded to three fluorine atoms, with two lone pairs of electrons on the chlorine atom.
Why is ClF3 highly reactive?
ClF3 is highly reactive due to its electron-deficient chlorine atom and the presence of highly electronegative fluorine atoms. This combination results in a molecule that readily reacts with other substances.
Conclusion and Next Steps
In conclusion, understanding the Lewis diagram for ClF3 provides valuable insights into its molecular structure, bonding, and properties. From its T-shaped geometry to its polar nature, ClF3's characteristics make it a fascinating subject of study in chemistry. Whether used in industrial applications or studied in academic settings, ClF3 exemplifies the complexities and wonders of chemical bonding.
We encourage readers to explore further resources on ClF3 and related topics. For more information, refer to authoritative sources such as the National Center for Biotechnology Information and the International Union of Pure and Applied Chemistry.
Feel free to leave a comment or share this article with others who might find it useful. Additionally, explore other articles on our website to deepen your understanding of chemistry and related fields.



