Lewis Dot of ClF3: Understanding Its Structure and Importance
ClF3, or Chlorine Trifluoride, is a fascinating molecule with a unique Lewis dot structure that plays a crucial role in chemistry. Understanding its structure helps us delve into the world of chemical bonding and molecular geometry. The Lewis dot of ClF3 is an essential concept for students and professionals alike who wish to explore the intricacies of molecular science.
The exploration of the Lewis dot structure of ClF3 is more than just academic. It is a practical application of theoretical chemistry that has implications in various industries. From materials science to environmental studies, understanding the Lewis dot of ClF3 provides insights into how molecules interact and form complex compounds.
In this comprehensive article, we will explore the intricacies of the Lewis dot structure of ClF3. We will break down its components, analyze its significance, and uncover the reasons why this molecule is of great interest to scientists and researchers. Let’s dive into the world of ClF3 and uncover its secrets.
What is ClF3 and Why is it Important?
ClF3, also known as Chlorine Trifluoride, is a highly reactive and toxic chemical compound. It is primarily used in the semiconductor industry for etching materials and in the production of uranium hexafluoride for nuclear fuel processing. Understanding its Lewis dot structure is crucial because it helps explain the molecule's behavior and reactivity.
ClF3 is a molecule with a T-shaped geometry, which arises from its unique arrangement of atoms. This structure plays a vital role in determining its chemical properties and interactions. By examining the Lewis dot structure, we can gain insights into why ClF3 is such a potent oxidizing agent.
Understanding the Lewis Dot Structure
Definition and Basics
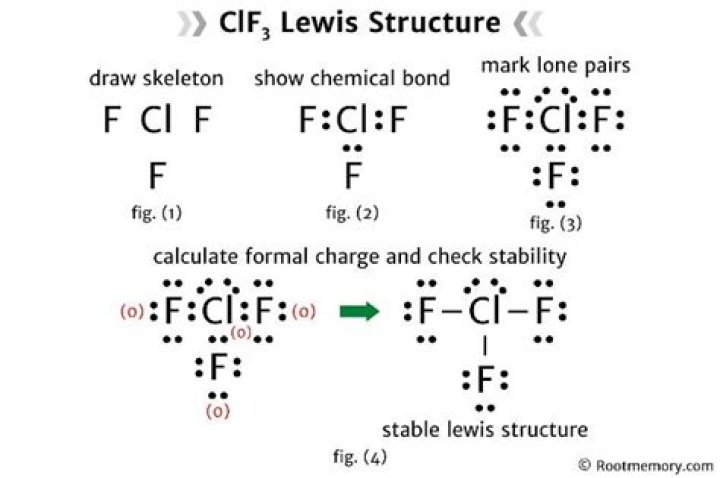
The Lewis dot structure is a diagram that represents the valence electrons of atoms in a molecule. It helps visualize how atoms bond with each other and the arrangement of electrons in the molecule. For ClF3, the Lewis dot structure shows how chlorine (Cl) bonds with three fluorine (F) atoms, with two lone pairs of electrons on the chlorine atom.
- Chlorine has seven valence electrons.
- Each fluorine atom has seven valence electrons.
- In total, ClF3 has 28 valence electrons.
Steps to Draw the Lewis Dot of ClF3
Step 1: Determine the Central Atom
In ClF3, chlorine is the central atom because it is less electronegative than fluorine. This is a fundamental principle when drawing Lewis dot structures. Chlorine will be the atom to which the fluorine atoms bond.
Step 2: Calculate Total Valence Electrons
To draw the Lewis dot structure, we need to calculate the total number of valence electrons in the molecule. For ClF3, this can be done as follows:
- Chlorine contributes 7 electrons.
- Each fluorine contributes 7 electrons, and there are three fluorine atoms.
- Total = 7 + (7 x 3) = 28 valence electrons.
Step 3: Place Lone Pairs and Bonds
Once the central atom and total valence electrons are determined, the next step is to place lone pairs and bonds. Chlorine forms single bonds with each fluorine atom, and the remaining electrons are placed as lone pairs on the chlorine atom.
ClF3 Molecular Geometry
T-shaped Structure
The molecular geometry of ClF3 is T-shaped due to the presence of two lone pairs on the chlorine atom. These lone pairs repel the bonding pairs of electrons, resulting in a unique geometry that is different from other molecules with similar formulas. The T-shaped structure is essential in understanding the reactivity and properties of ClF3.
Electron Pair Geometry vs Molecular Geometry
Understanding the Difference
While the electron pair geometry of ClF3 is trigonal bipyramidal, its molecular geometry is T-shaped. This difference arises because the lone pairs on the chlorine atom influence the arrangement of the bonding pairs. Understanding this distinction is crucial in predicting the behavior of molecules.
Properties of ClF3
Physical and Chemical Properties
ClF3 is a colorless gas at room temperature but can condense into a pale greenish-yellow liquid under pressure. It is highly reactive and can ignite combustible materials without an external ignition source. Some key properties include:
- Molecular Weight: 92.45 g/mol
- Boiling Point: 11.78°C
- Melting Point: -76.3°C
Applications of ClF3
Industrial Uses
ClF3 finds applications in several industries due to its unique properties. Some of the most notable uses include:
- Semiconductor Manufacturing: Used for etching silicon wafers.
- Nuclear Industry: Employed in the production of uranium hexafluoride.
- Propellants: Utilized in rocket propellants due to its high oxidizing potential.
Safety Considerations
Handling ClF3 Safely
Due to its highly reactive and toxic nature, ClF3 requires careful handling. Safety measures include:
- Using proper personal protective equipment (PPE).
- Working in well-ventilated areas or fume hoods.
- Following strict safety protocols during storage and transportation.
Environmental Impact
Understanding the Effects
ClF3 can have significant environmental impacts due to its reactivity and toxicity. It can react violently with water and organic materials, releasing harmful gases. Proper disposal and containment are essential to minimize its environmental footprint.
Conclusion
The Lewis dot structure of ClF3 is a fascinating topic that combines theoretical chemistry with practical applications. By understanding its structure, we gain insights into its reactivity, properties, and uses. ClF3 is a powerful molecule with applications in various industries, but its handling requires caution due to its toxic and reactive nature.
We encourage readers to explore further and deepen their understanding of molecular chemistry. Share your thoughts and questions in the comments below, and don’t forget to check out other articles on our website for more insights into the world of chemistry.
References
The information in this article is based on reliable sources, including:



