Lewis Structure for Chlorine Trifluoride: A Comprehensive Guide
Understanding the Lewis structure for chlorine trifluoride (ClF₃) is essential for anyone studying chemistry, especially in the context of molecular geometry and chemical bonding. This compound, known for its unique T-shaped molecular structure, plays a significant role in understanding the behavior of interhalogen compounds. By analyzing its Lewis structure, chemists can predict the compound's reactivity, stability, and interactions with other substances.
Chlorine trifluoride is a fascinating molecule due to its ability to form strong bonds while maintaining a distinct molecular shape. In this article, we will delve into the intricacies of ClF₃'s Lewis structure, exploring its formation, characteristics, and implications in chemical reactions. Whether you're a student, researcher, or simply curious about molecular structures, this guide will provide valuable insights into ClF₃'s properties.
Our exploration will cover essential topics such as electron distribution, bond angles, and molecular geometry. Additionally, we will discuss the significance of the Lewis structure in predicting the behavior of chlorine trifluoride in various chemical environments. Let's begin by examining the foundational aspects of this remarkable molecule.
Introduction to Lewis Structure
The Lewis structure is a fundamental concept in chemistry that represents the arrangement of valence electrons in a molecule. By visualizing these electrons, chemists can predict the molecule's shape, polarity, and reactivity. For chlorine trifluoride (ClF₃), the Lewis structure provides critical insights into its unique T-shaped geometry and the distribution of electrons around the central chlorine atom.
Why Lewis Structures Matter
Lewis structures are essential tools for understanding molecular bonding. They allow chemists to visualize how atoms share electrons and form stable configurations. In the case of ClF₃, the structure reveals the presence of lone pairs and bonded pairs, which significantly influence the molecule's geometry and properties.
Basics of Lewis Structures
A Lewis structure is a two-dimensional representation of a molecule that shows how valence electrons are distributed among its atoms. To construct a Lewis structure, chemists follow specific rules:
- Determine the total number of valence electrons in the molecule.
- Identify the central atom and arrange the surrounding atoms.
- Distribute electrons to satisfy the octet rule for each atom.
- Account for any formal charges if necessary.
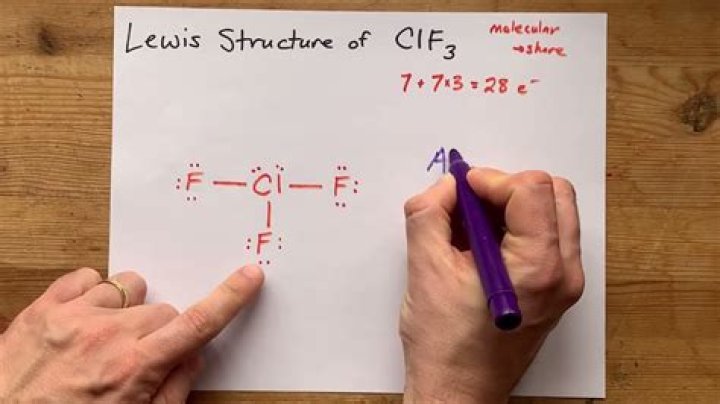
For ClF₃, the central chlorine atom forms three single bonds with fluorine atoms, leaving two lone pairs of electrons on the chlorine atom.
Overview of Chlorine Trifluoride
Chlorine trifluoride (ClF₃) is a highly reactive interhalogen compound consisting of one chlorine atom and three fluorine atoms. It was first synthesized in 1936 and has since been used in various industrial applications, including rocket propellants and chemical manufacturing processes.
Key Properties of ClF₃
ClF₃ is a colorless gas at room temperature but can condense into a pale green liquid under pressure. It is highly toxic and corrosive, requiring careful handling in laboratory and industrial settings. The molecule's reactivity stems from its electron distribution and molecular geometry, which we will explore in detail.
Electron Distribution in ClF₃
In the Lewis structure of ClF₃, the central chlorine atom contributes seven valence electrons, while each fluorine atom contributes seven valence electrons. This results in a total of 28 valence electrons for the molecule. These electrons are distributed as follows:
- Three single bonds between chlorine and fluorine atoms (six electrons).
- Two lone pairs on the chlorine atom (four electrons).
- Remaining electrons satisfy the octet rule for each fluorine atom.
This distribution leads to the formation of a T-shaped molecular geometry, as the lone pairs on chlorine repel the bonding pairs, causing the fluorine atoms to occupy positions that minimize repulsion.
Molecular Geometry
The molecular geometry of ClF₃ is determined by the VSEPR (Valence Shell Electron Pair Repulsion) theory, which predicts the arrangement of electron pairs around the central atom. In ClF₃, the two lone pairs on chlorine cause significant repulsion, resulting in a T-shaped geometry rather than a trigonal bipyramidal shape.
Factors Influencing Geometry
Several factors contribute to ClF₃'s unique geometry:
- The presence of lone pairs on the central atom.
- The electronegativity difference between chlorine and fluorine.
- The minimization of electron pair repulsion according to VSEPR theory.
Understanding these factors is crucial for predicting the behavior of ClF₃ in chemical reactions and its interactions with other substances.
Bond Angles
The bond angles in ClF₃ are slightly less than 90° due to the repulsion between the lone pairs and bonding pairs of electrons. This deviation from the ideal geometry is a result of the lone pair-bond pair repulsion, which is stronger than bond pair-bond pair repulsion. The exact bond angles can be calculated using advanced computational methods, but experimental data suggests angles of approximately 87.5° between the axial and equatorial fluorine atoms.
Significance of Bond Angles
The bond angles in ClF₃ have important implications for its chemical properties:
- They determine the molecule's overall shape and polarity.
- They influence the molecule's reactivity with other substances.
- They provide insights into the stability of the molecule under different conditions.
By understanding these angles, chemists can better predict ClF₃'s behavior in various chemical environments.
Resonance Structures
While ClF₃ does not exhibit classical resonance structures, the distribution of electrons around the central chlorine atom can be analyzed using formal charge considerations. The lone pairs on chlorine contribute to the molecule's stability, ensuring that the bonding configuration remains consistent with the octet rule.
Implications for Stability
The absence of resonance structures in ClF₃ highlights the importance of electron distribution in maintaining molecular stability. The lone pairs on chlorine play a critical role in minimizing repulsion and ensuring the molecule's overall stability. This stability is essential for ClF₃'s applications in various chemical processes.
Applications of ClF₃
Chlorine trifluoride finds applications in several industries due to its unique chemical properties:
- Rocket Propellants: ClF₃ is used as an oxidizer in rocket fuel due to its high reactivity and ability to release large amounts of energy.
- Chemical Manufacturing: It serves as a fluorinating agent in the production of various fluorinated compounds.
- Material Processing: ClF₃ is employed in the etching of materials, particularly in semiconductor manufacturing.
Despite its usefulness, ClF₃ requires careful handling due to its toxicity and corrosiveness.
Safety Considerations
Handling ClF₃ requires strict adherence to safety protocols due to its hazardous nature. The compound is highly reactive, toxic, and corrosive, posing significant risks to human health and the environment. Safety measures include:
- Using personal protective equipment (PPE) such as gloves, goggles, and respirators.
- Working in well-ventilated areas or fume hoods.
- Storing ClF₃ in compatible containers away from moisture and other reactive substances.
By following these precautions, researchers and industrial workers can safely handle ClF₃ while minimizing risks.
Conclusion
The Lewis structure of chlorine trifluoride provides valuable insights into its molecular geometry, electron distribution, and chemical properties. By understanding the T-shaped geometry and bond angles of ClF₃, chemists can predict its behavior in various applications, from rocket propellants to semiconductor manufacturing. However, due to its toxicity and reactivity, ClF₃ must be handled with extreme caution.
We encourage readers to explore further resources on molecular structures and chemical bonding to deepen their understanding of this fascinating compound. Please leave your thoughts and questions in the comments section below, and consider sharing this article with others who may find it informative. For more chemistry-related content, explore our other articles and resources.
References:
- Brown, T. L., LeMay, H. E., Bursten, B. E., & Murphy, C. J. (2014). Chemistry: The Central Science. Pearson Education.
- Housecroft, C. E., & Sharpe, A. G. (2012). Inorganic Chemistry. Pearson Education.
- Moore, J. W., Stanitski, C. L., & Jurs, P. C. (2009). Chemistry: The Molecular Science. Cengage Learning.



