Lewis Structure of CIF3: Understanding the Chemistry Behind This Fascinating Compound
When it comes to understanding chemical compounds, the Lewis structure of CIF3 plays a crucial role in determining its properties and behavior. CIF3, or chlorine trifluoride, is an intriguing molecule that has unique characteristics and applications in various industries. By exploring the Lewis structure of CIF3, we gain insight into its molecular geometry, bond formation, and reactivity. This article will delve into the fascinating world of CIF3 and provide a comprehensive understanding of its Lewis structure.
Chemistry enthusiasts and students often find themselves captivated by the intricate nature of molecular structures. CIF3, with its unusual shape and properties, offers an excellent opportunity to explore the principles of chemical bonding and molecular geometry. Understanding the Lewis structure of CIF3 not only enhances our knowledge of chemistry but also has practical implications in fields such as materials science and chemical engineering.
In this article, we will dissect the Lewis structure of CIF3, discussing its formation, stability, and significance. By the end of this exploration, you will have a clear understanding of why CIF3 behaves the way it does and how its structure influences its chemical properties. Let's dive into the world of CIF3 and uncover its secrets.
Introduction to CIF3
What is CIF3?
CIF3, or chlorine trifluoride, is a highly reactive compound that consists of one chlorine atom and three fluorine atoms. It is a colorless gas at room temperature but can appear yellowish under certain conditions. CIF3 is primarily used in the semiconductor industry for etching materials and in the production of uranium hexafluoride (UF6) for nuclear fuel processing.
One of the most interesting aspects of CIF3 is its ability to react with almost any substance it comes into contact with, making it both useful and dangerous in industrial applications. Its reactivity is largely due to its unique Lewis structure and molecular geometry, which we will explore in detail below.
Lewis Structure Definition
A Lewis structure is a diagram that shows the bonding between atoms in a molecule and the lone pairs of electrons that may exist in the molecule. It is named after Gilbert N. Lewis, who introduced the concept in 1916. The Lewis structure helps chemists predict the geometry of molecules, understand their reactivity, and determine their stability.
In the case of CIF3, the Lewis structure provides valuable insights into how the chlorine and fluorine atoms interact to form this highly reactive compound. By examining the electron distribution and bond angles, we can better understand the properties of CIF3.
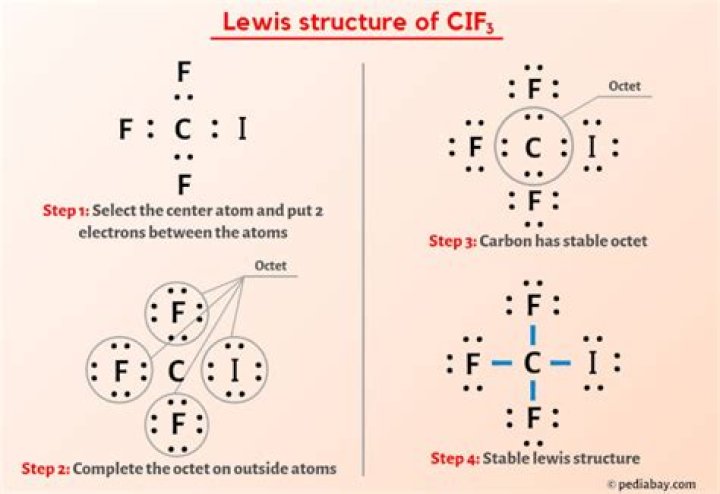
Drawing the Lewis Structure of CIF3
Step-by-Step Guide to Drawing CIF3
Drawing the Lewis structure of CIF3 involves a systematic approach to ensure accuracy. Here's how you can do it:
- Count the total number of valence electrons: Chlorine has 7 valence electrons, and each fluorine atom contributes 7 valence electrons. Therefore, the total number of valence electrons in CIF3 is 28.
- Place the chlorine atom in the center and surround it with the three fluorine atoms.
- Distribute the electrons around each atom to satisfy the octet rule. Chlorine will have three single bonds with the fluorine atoms and two lone pairs of electrons.
- Check the formal charges to ensure the most stable arrangement of electrons.
Molecular Geometry of CIF3
T-shaped Geometry
The molecular geometry of CIF3 is T-shaped due to the presence of two lone pairs of electrons on the chlorine atom. These lone pairs repel the bonding pairs of electrons, causing the three fluorine atoms to arrange themselves in a T-shape around the chlorine atom. This arrangement minimizes electron repulsion and results in a stable molecular structure.
The bond angles in CIF3 are approximately 90° between the axial fluorine atoms and the equatorial fluorine atom. This geometry is a direct consequence of the VSEPR (Valence Shell Electron Pair Repulsion) theory, which predicts the shape of molecules based on the repulsion between electron pairs.
Bonding in CIF3
Covalent Bonds and Electronegativity
The bonding in CIF3 is covalent, with chlorine and fluorine sharing electrons to form stable bonds. Fluorine, being the most electronegative element, pulls the shared electrons closer to itself, creating a polar bond. This polarity contributes to the high reactivity of CIF3, as the partial positive charge on the chlorine atom makes it more susceptible to nucleophilic attack.
In addition to the three sigma bonds between chlorine and fluorine, CIF3 also exhibits pi-bond characteristics due to the overlap of p orbitals. This overlap strengthens the bonds and contributes to the overall stability of the molecule.
Stability of CIF3
Factors Affecting Stability
The stability of CIF3 is influenced by several factors, including its electron distribution, molecular geometry, and bond strength. While CIF3 is highly reactive, its stability is maintained by the strong covalent bonds between chlorine and fluorine. The presence of lone pairs on the chlorine atom also contributes to its stability, as they help balance the electron density around the molecule.
However, CIF3 is thermodynamically unstable and can decompose into chlorine and fluorine gases at high temperatures. This instability is a result of its high reactivity and the energy required to break the covalent bonds.
Applications of CIF3
Industrial Uses
CIF3 finds applications in various industries due to its unique properties. Some of its key applications include:
- Semiconductor manufacturing: CIF3 is used as an etchant for silicon and other materials in the production of microchips.
- Nuclear fuel processing: CIF3 is employed in the conversion of uranium dioxide (UO2) to uranium hexafluoride (UF6) for enrichment processes.
- Chemical research: CIF3 serves as a powerful oxidizing and fluorinating agent in laboratory settings.
Despite its utility, the handling of CIF3 requires extreme caution due to its high reactivity and potential hazards.
Common Misconceptions About CIF3
Clearing the Air
There are several misconceptions surrounding CIF3 that need clarification:
- CIF3 is not a naturally occurring compound; it is synthesized in laboratories and industrial settings.
- Although CIF3 is highly reactive, it is not inherently unstable under normal conditions.
- The reactivity of CIF3 does not make it dangerous in all contexts; proper handling and safety measures mitigate risks.
Comparison of CIF3 with Other Compounds
Similarities and Differences
When compared to other halogen compounds, CIF3 exhibits both similarities and differences. For instance:
- Like other halogen compounds, CIF3 forms covalent bonds and exhibits high reactivity.
- Unlike chlorine trifluoride, bromine trifluoride (BrF3) has a square pyramidal geometry due to the presence of three lone pairs on the bromine atom.
- CIF3 is less stable than some other halogen compounds, such as chlorine trifluoride (ClF3), due to its smaller size and higher electronegativity.
Future Research Directions for CIF3
Exploring New Horizons
Future research on CIF3 could focus on:
- Developing safer methods for handling and transporting CIF3 in industrial applications.
- Investigating new uses for CIF3 in emerging technologies, such as quantum computing and advanced materials.
- Studying the environmental impact of CIF3 and devising strategies to minimize its ecological footprint.
Conclusion
In conclusion, the Lewis structure of CIF3 provides valuable insights into the molecular geometry, bonding, and reactivity of this fascinating compound. By understanding the principles behind its structure, we can better appreciate its applications and potential risks. As we continue to explore the world of chemistry, CIF3 remains a compelling subject of study, offering opportunities for innovation and discovery.
We invite you to share your thoughts and questions in the comments section below. Additionally, feel free to explore other articles on our site for more in-depth discussions on various scientific topics. Together, let's continue to expand our knowledge and understanding of the world around us.