Lewis Structure of Methane: A Comprehensive Guide
Methane (CH₄) is one of the simplest hydrocarbons and plays a vital role in chemistry, biology, and environmental science. Understanding its Lewis structure is fundamental to comprehending its chemical properties and reactivity. If you're diving into organic chemistry or environmental studies, mastering the Lewis structure of methane is essential.
Methane, often referred to as natural gas, is the primary component of fossil fuels. Its molecular structure provides insight into its stability and its role as a greenhouse gas. By understanding its Lewis structure, you can better appreciate its behavior in various chemical reactions.
In this article, we'll delve into the intricacies of the Lewis structure of methane, exploring its formation, properties, and significance. Whether you're a student, researcher, or simply curious about chemistry, this guide will provide you with all the necessary information.
Introduction to Lewis Structures
A Lewis structure is a diagram that represents the bonding between atoms in a molecule and the lone pairs of electrons. It helps chemists visualize the arrangement of electrons in a molecule, which is crucial for understanding its properties and reactivity.
For methane (CH₄), the Lewis structure is particularly simple yet highly informative. By examining the electron distribution, we can predict the molecule's geometry, polarity, and other characteristics.
Understanding Lewis structures is foundational for studying more complex molecules and reactions. This section will provide an overview of the principles behind Lewis structures and their importance in chemistry.
Overview of Methane
What is Methane?
Methane is the simplest alkane and the first member of the hydrocarbon series. It consists of one carbon atom bonded to four hydrogen atoms. Methane is colorless, odorless, and highly flammable, making it a valuable energy source.
Chemically, methane is represented as CH₄. Its molecular weight is approximately 16.04 g/mol, and it exists as a gas under standard conditions.
Significance of Methane
Methane is not only important as a fuel but also as a precursor to many industrial chemicals. Additionally, it plays a significant role in the Earth's atmosphere, contributing to the greenhouse effect.
- Methane is a primary component of natural gas.
- It is used in the production of hydrogen, methanol, and other chemicals.
- Methane emissions are a major concern in climate change discussions.
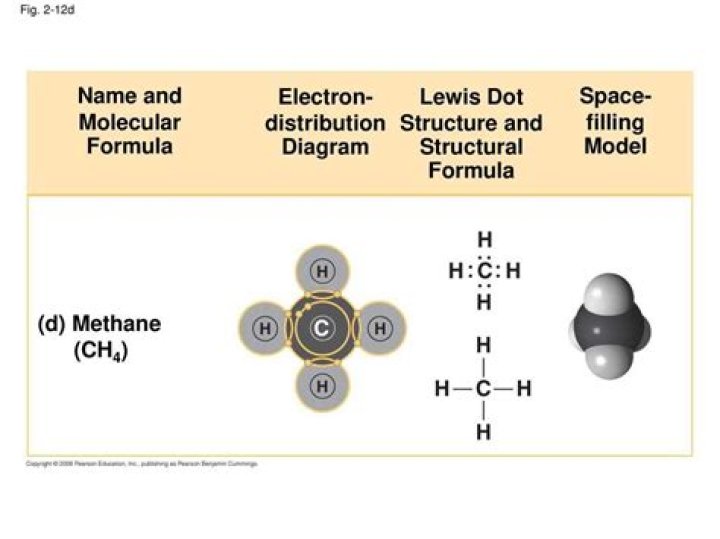
Lewis Structure of Methane
The Lewis structure of methane involves a central carbon atom surrounded by four hydrogen atoms. Each hydrogen atom shares one electron with the carbon atom, forming four single covalent bonds.
Here’s how the Lewis structure of methane is constructed:
- Carbon has four valence electrons, while each hydrogen atom has one.
- These electrons are shared to form four single bonds, satisfying the octet rule for carbon.
- Each hydrogen atom achieves a stable duet configuration.
This simple yet elegant structure explains the stability and reactivity of methane.
Molecular Geometry and Bond Angles
Tetrahedral Geometry
The molecular geometry of methane is tetrahedral. This shape arises due to the equal repulsion between the four electron pairs around the carbon atom. The bond angles in methane are approximately 109.5°, which is characteristic of tetrahedral geometry.
This geometry ensures that the molecule is nonpolar, as the electronegativity difference between carbon and hydrogen is negligible.
Implications of Tetrahedral Shape
The tetrahedral shape of methane has significant implications for its chemical behavior:
- It determines the molecule's stability and resistance to reactions.
- The geometry influences how methane interacts with other molecules.
- Understanding this shape is crucial for predicting reaction mechanisms.
Hybridization in Methane
In methane, the carbon atom undergoes sp³ hybridization. This process involves the mixing of one 2s orbital and three 2p orbitals to form four equivalent sp³ hybrid orbitals. These orbitals then overlap with the 1s orbitals of the hydrogen atoms to form four σ bonds.
Sp³ hybridization explains the tetrahedral geometry and the equal bond lengths in methane. Each C-H bond has a length of approximately 1.09 Å, contributing to the molecule's stability.
Properties of Methane
Physical Properties
Methane exhibits several notable physical properties:
- Boiling Point: -161.5°C
- Melting Point: -182.5°C
- Density: 0.717 kg/m³
Its low boiling and melting points are due to its small size and nonpolar nature.
Chemical Properties
Methane is relatively inert under standard conditions but can undergo combustion and substitution reactions. When burned in the presence of oxygen, it produces carbon dioxide and water:
CH₄ + 2O₂ → CO₂ + 2H₂O
Substitution reactions, such as chlorination, can also occur under certain conditions:
CH₄ + Cl₂ → CH₃Cl + HCl
Applications of Methane
Methane has numerous applications across various industries:
- Energy Production: Methane is a primary component of natural gas, used for heating and electricity generation.
- Chemical Synthesis: It serves as a raw material for producing hydrogen, ammonia, and methanol.
- Transportation Fuel: Compressed natural gas (CNG) and liquefied natural gas (LNG) are used as alternative fuels for vehicles.
Its versatility makes methane an indispensable resource in modern society.
Environmental Impact of Methane
Methane is a potent greenhouse gas, with a global warming potential 25 times greater than carbon dioxide over a 100-year period. It contributes significantly to climate change through:
- Agricultural activities, such as livestock farming.
- Fossil fuel extraction and distribution.
- Landfills and wastewater treatment.
Mitigating methane emissions is a critical step in addressing global warming.
Frequently Asked Questions
What is the Lewis structure of methane?
The Lewis structure of methane consists of a central carbon atom bonded to four hydrogen atoms via single covalent bonds. Each atom achieves a stable electron configuration through this arrangement.
Why is methane nonpolar?
Methane is nonpolar because of its symmetrical tetrahedral geometry and the similar electronegativity of carbon and hydrogen atoms. This symmetry ensures that the dipole moments cancel out.
How does methane contribute to global warming?
Methane traps heat in the atmosphere more effectively than carbon dioxide. Its release from sources like agriculture, fossil fuels, and landfills exacerbates the greenhouse effect, contributing to climate change.
Conclusion
The Lewis structure of methane provides valuable insights into its chemical properties and behavior. By understanding its molecular geometry, hybridization, and physical properties, we can better appreciate its role in energy production and environmental science.
We encourage you to explore further resources on methane and its impact on our planet. Feel free to leave comments or questions below, and don't forget to share this article with others who may find it useful.
References:
- Chemistry: The Central Science by Brown, LeMay, and Bursten
- Environmental Science by Cunningham and Cunningham
- International Panel on Climate Change (IPCC) Reports