One End of a Battery: Understanding Its Role and Function
Batteries are an essential part of modern life, powering everything from smartphones to cars. One end of a battery plays a critical role in its functionality, and understanding how it works can help you make better decisions about your devices and energy consumption. In this article, we will explore the science behind batteries, focusing on the importance of one end of a battery and how it impacts performance.
Batteries have become indispensable in our daily lives, powering countless devices that we rely on. Whether it's your phone, laptop, or even your car, batteries are the backbone of modern technology. However, many people don't fully understand how batteries work or the significance of their components. This article aims to demystify the role of one end of a battery and explain why it's so crucial to the overall function of the device.
By the end of this article, you will have a deeper understanding of battery technology, specifically the role of one end of a battery, and how it contributes to the energy transfer process. Whether you're a tech enthusiast or simply curious about how your devices work, this guide will provide valuable insights into the science of batteries.
What Is One End of a Battery?
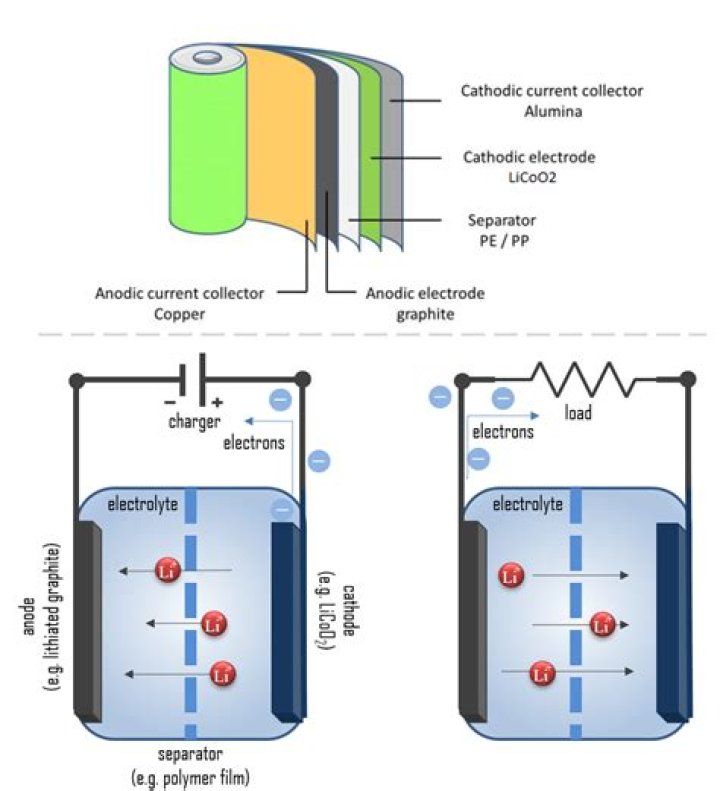
When discussing batteries, it's important to understand the basic structure and components. One end of a battery refers to either the positive or negative terminal, which plays a vital role in the flow of electrical current. The positive terminal is typically marked with a "+" sign, while the negative terminal is marked with a "-" sign. These terminals are the points of contact where energy is transferred to and from the device.
Understanding Battery Terminals
The terminals of a battery are essential for its operation. The positive terminal, also known as the cathode, is where oxidation occurs, releasing electrons. The negative terminal, or anode, is where reduction takes place, accepting electrons. This process creates a flow of electrical current that powers devices.
- Positive Terminal (Cathode): Responsible for releasing electrons
- Negative Terminal (Anode): Responsible for accepting electrons
- Both terminals work together to create a complete circuit
How Does One End of a Battery Work?
The function of one end of a battery is closely tied to the electrochemical reactions that occur within the battery. When a device is connected to the battery, a chemical reaction is initiated at the anode, releasing electrons. These electrons flow through the device, providing power, and then return to the cathode, completing the circuit.
Electrochemical Reactions in Batteries
The electrochemical reactions in batteries involve the transfer of ions between the anode and cathode. This process generates electrical energy, which is used to power devices. Understanding these reactions is key to grasping how one end of a battery contributes to the overall functionality of the device.
- Anode: Loses electrons during oxidation
- Cathode: Gains electrons during reduction
- Electrolyte: Facilitates the movement of ions between terminals
Types of Batteries and Their Terminals
There are various types of batteries available, each with its own unique characteristics. The design and materials used in batteries can affect the performance of one end of a battery. Some common types of batteries include alkaline, lithium-ion, and nickel-metal hydride batteries.
Alkaline Batteries
Alkaline batteries are widely used in household devices and are known for their long shelf life and reliable performance. The terminals of alkaline batteries are designed to provide a steady flow of electrical current, making them ideal for low-drain devices.
Lithium-Ion Batteries
Lithium-ion batteries are commonly found in smartphones, laptops, and electric vehicles. These batteries are rechargeable and offer high energy density. The terminals of lithium-ion batteries are engineered to handle high current loads, making them suitable for power-hungry devices.
The Importance of One End of a Battery
The significance of one end of a battery cannot be overstated. Without properly functioning terminals, a battery cannot deliver the necessary power to a device. The design and quality of the terminals directly impact the efficiency and lifespan of the battery.
Factors Affecting Terminal Performance
Several factors can influence the performance of one end of a battery, including:
- Material quality: High-quality materials improve conductivity and durability
- Corrosion: Preventing corrosion is crucial for maintaining terminal integrity
- Design: Proper design ensures optimal contact with the device
Common Issues with Battery Terminals
Despite their importance, battery terminals can experience issues that affect performance. Corrosion, poor contact, and damage are some of the most common problems. Addressing these issues promptly can help extend the life of your battery and improve device performance.
Preventing Terminal Corrosion
Corrosion is a common problem with battery terminals, especially in environments with high humidity or temperature fluctuations. To prevent corrosion:
- Use anti-corrosion sprays or grease
- Keep terminals clean and dry
- Regularly inspect and maintain terminals
Advancements in Battery Technology
As technology continues to evolve, so does battery design. Researchers are constantly exploring new materials and methods to improve the efficiency and longevity of batteries. These advancements often focus on enhancing the performance of one end of a battery, leading to better overall results.
Innovations in Terminal Design
New innovations in terminal design include the use of advanced materials and coatings to improve conductivity and reduce corrosion. These improvements not only enhance battery performance but also contribute to a more sustainable future by increasing the lifespan of batteries.
Safety Considerations for Battery Terminals
While batteries are essential for modern technology, they can pose safety risks if not handled properly. One end of a battery, in particular, requires careful attention to prevent accidents. Short circuits, overheating, and leakage are potential hazards that can arise from improper handling or damage to the terminals.
Best Practices for Battery Safety
To ensure safe use of batteries, follow these best practices:
- Always handle batteries with care
- Avoid exposing batteries to extreme temperatures
- Dispose of batteries properly to prevent environmental damage
Environmental Impact of Batteries
Batteries have a significant environmental impact, particularly in terms of waste management and resource consumption. Proper disposal and recycling of batteries are essential to minimize their impact on the environment. Understanding the role of one end of a battery can also contribute to more sustainable practices in battery design and usage.
Recycling Batteries
Recycling batteries not only reduces waste but also recovers valuable materials that can be reused in new batteries. Many organizations offer battery recycling programs, making it easier for consumers to dispose of their batteries responsibly.
Conclusion
In conclusion, one end of a battery plays a crucial role in the functionality and performance of the device. Understanding the science behind battery terminals and their importance can help you make informed decisions about your devices and energy consumption. By following best practices for battery care and safety, you can extend the life of your batteries and reduce their environmental impact.
We encourage you to share this article with others who may benefit from the information. If you have any questions or comments, feel free to leave them below. Additionally, explore our other articles for more insights into technology and sustainability.
Table of Contents
How Does One End of a Battery Work?
Types of Batteries and Their Terminals
The Importance of One End of a Battery
Common Issues with Battery Terminals
Advancements in Battery Technology
Safety Considerations for Battery Terminals
Environmental Impact of Batteries
References:
- U.S. Department of Energy - Battery Technologies
- National Renewable Energy Laboratory - Battery Research
- Environmental Protection Agency - Battery Recycling


