One End of a Battery: Understanding the Science, Uses, and Safety
Have you ever wondered what happens at one end of a battery? Whether it's powering your smartphone, remote control, or electric car, understanding the role of one end of a battery is crucial for maximizing its efficiency and ensuring safety. In this article, we will delve into the fascinating world of batteries, exploring their components, functions, and applications.
Batteries are indispensable in modern life, and understanding their inner workings can help you make informed decisions about energy usage. By focusing on one end of a battery, we uncover the science behind how electrical energy is generated and delivered to our devices.
This article will explore the importance of one end of a battery in detail, offering practical insights and actionable tips. Whether you're a tech enthusiast, a student, or simply curious about how batteries work, this guide will provide you with comprehensive knowledge.
Introduction to One End of a Battery
When we talk about one end of a battery, we are referring to either the positive or negative terminal, each playing a vital role in the battery's operation. These terminals are the points of contact where electrical energy is transferred to and from the battery.
The positive terminal, often marked with a "+" sign, is the source of electrical current that flows out of the battery to power devices. On the other hand, the negative terminal, marked with a "-" sign, completes the circuit by allowing current to flow back into the battery. Understanding the functions of these terminals is essential for optimizing battery performance.
In this section, we will explore the basic principles of how one end of a battery operates and its significance in various applications. By gaining a deeper understanding of these components, we can better appreciate the technology that powers our daily lives.
Battery Anatomy: Understanding the Structure
Components of a Battery
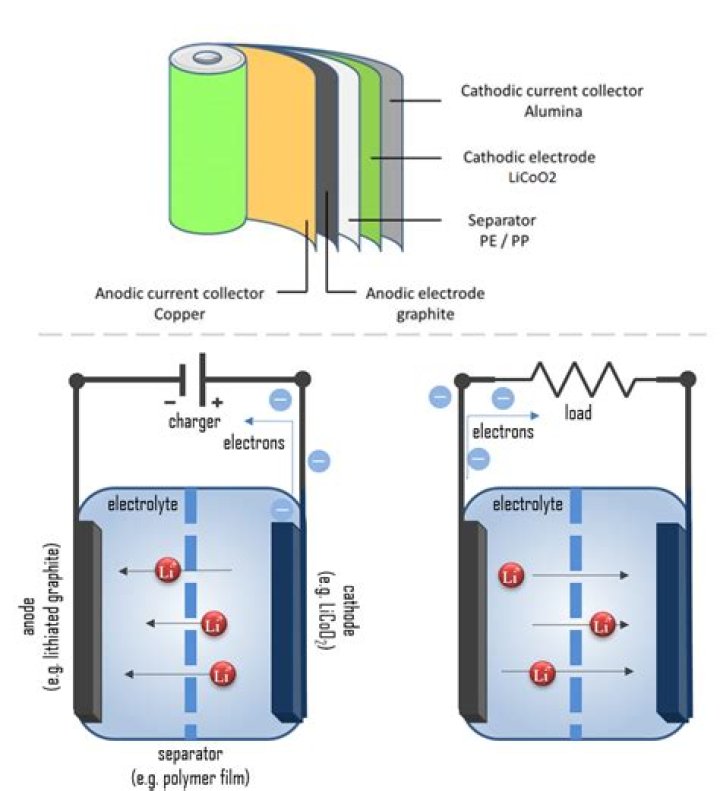
A battery is composed of several key components, each contributing to its functionality. The primary parts include the anode, cathode, electrolyte, separator, and terminals. The anode and cathode are the electrodes where the chemical reactions occur, while the electrolyte facilitates ion movement between them.
- Anode: The negative electrode where oxidation occurs.
- Cathode: The positive electrode where reduction takes place.
- Electrolyte: A substance that allows the flow of ions between the anode and cathode.
- Separator: A barrier that prevents direct contact between the anode and cathode while allowing ion exchange.
- Terminals: The points of contact for external circuits.
The Positive Terminal: What It Does
The positive terminal of a battery, also known as the cathode, is where the electrical current flows out to power devices. This terminal is essential for initiating the flow of electrons that drives the electrical circuit. When connected to a device, the positive terminal ensures that the energy stored in the battery is effectively utilized.
Understanding the role of the positive terminal is crucial for optimizing battery performance. For instance, ensuring proper contact between the terminal and the device can prevent energy loss and improve efficiency.
The Negative Terminal: Its Role and Function
At the other end of the battery lies the negative terminal, or anode, which plays a complementary role to the positive terminal. The negative terminal is responsible for receiving the flow of electrons back into the battery, completing the circuit. This process is essential for maintaining the battery's functionality and ensuring a continuous supply of energy.
Proper maintenance of the negative terminal is vital for preventing issues such as corrosion and ensuring long-term battery life. Regular cleaning and inspection can help mitigate these problems.
Energy Flow: How One End of a Battery Works
Chemical Reactions in Batteries
The operation of one end of a battery relies on a series of chemical reactions that convert stored chemical energy into electrical energy. At the positive terminal, reduction occurs, where ions gain electrons, while oxidation takes place at the negative terminal, where ions lose electrons. This exchange of electrons generates the electrical current that powers devices.
Understanding these chemical processes is essential for grasping the mechanics of battery operation. By studying the interactions between the anode, cathode, and electrolyte, scientists and engineers can develop more efficient and sustainable battery technologies.
Types of Batteries and Their Applications
Primary vs. Secondary Batteries
Batteries can be broadly categorized into two types: primary and secondary. Primary batteries, such as alkaline batteries, are designed for single-use and cannot be recharged. They are commonly used in devices like remote controls and flashlights. Secondary batteries, also known as rechargeable batteries, can be recharged multiple times and are widely used in smartphones, laptops, and electric vehicles.
- Alkaline Batteries: Known for their long shelf life and reliability.
- Lithium-Ion Batteries: Popular for their high energy density and rechargeability.
- Lead-Acid Batteries: Commonly used in automotive applications.
Safety Tips for Handling One End of a Battery
Handling one end of a battery requires caution to avoid potential hazards such as short circuits, overheating, and leakage. Here are some safety tips to keep in mind:
- Avoid Short Circuits: Ensure that the terminals do not come into contact with conductive materials.
- Store Properly: Keep batteries in a cool, dry place away from direct sunlight.
- Dispose Safely: Follow local regulations for recycling and disposing of batteries.
By following these guidelines, you can ensure the safe and effective use of batteries in your daily life.
Environmental Impact of Batteries
Sustainability and Recycling
The production and disposal of batteries have significant environmental implications. The extraction of raw materials such as lithium and cobalt can lead to habitat destruction and pollution. Moreover, improper disposal of batteries can result in toxic waste that harms ecosystems.
To mitigate these effects, efforts are being made to promote sustainable battery production and recycling. Governments and organizations are encouraging the development of eco-friendly battery technologies and establishing recycling programs to reduce waste.
Future Technologies: Innovations in Battery Design
The future of battery technology holds exciting possibilities. Researchers are exploring new materials and designs to improve energy storage capacity, efficiency, and sustainability. Solid-state batteries, for example, promise higher safety and longer lifespans compared to traditional lithium-ion batteries.
Additionally, advancements in nanotechnology and quantum mechanics are paving the way for breakthroughs in battery performance. These innovations have the potential to revolutionize industries ranging from consumer electronics to renewable energy.
Conclusion and Key Takeaways
In conclusion, understanding one end of a battery is essential for maximizing its performance and ensuring safe usage. From the basic anatomy of a battery to the chemical processes that drive its operation, this article has provided a comprehensive overview of the topic.
Key takeaways include the importance of proper terminal maintenance, the differences between primary and secondary batteries, and the environmental impact of battery production and disposal. By staying informed and adopting sustainable practices, we can contribute to a cleaner and more efficient energy future.
We invite you to share your thoughts and experiences in the comments section below. Additionally, feel free to explore other articles on our website for more insights into the world of energy and technology.
Sources:
- International Energy Agency (IEA) - Battery Storage Technology
- U.S. Department of Energy - Battery Research and Development
- Environmental Protection Agency (EPA) - Battery Recycling Guidelines



