Understanding CLF3 Geometry: A Comprehensive Guide
CLF3 geometry is a fascinating topic in chemistry that explores the molecular structure of chlorine trifluoride (CLF3). This compound plays a significant role in the field of chemistry due to its unique properties and applications. Understanding its geometry helps us grasp how atoms bond and interact within molecules, leading to specific physical and chemical behaviors. Whether you're a student, researcher, or simply curious about the world of chemistry, this article will provide an in-depth exploration of CLF3 geometry.
Chlorine trifluoride (CLF3) is a highly reactive compound used in various industrial applications, including rocket propellants and etching agents. Its molecular geometry is critical in determining its reactivity and functionality. By studying CLF3 geometry, we gain insights into the principles of molecular bonding and how these principles influence the compound's properties.
This article aims to provide a detailed understanding of CLF3 geometry, covering its structure, bond angles, hybridization, and applications. We will also explore how this knowledge can be applied in real-world scenarios, ensuring that readers leave with a comprehensive grasp of the subject.
Introduction to CLF3 Geometry
What is CLF3?
Chlorine trifluoride (CLF3) is a highly reactive interhalogen compound composed of one chlorine atom and three fluorine atoms. Its molecular geometry plays a crucial role in determining its chemical behavior. Understanding the geometry of CLF3 allows chemists to predict its reactivity and stability, making it an essential topic in advanced chemistry studies.
Why Study CLF3 Geometry?
The study of CLF3 geometry is vital for several reasons. First, it helps explain the compound's unique reactivity and its ability to act as a powerful oxidizing agent. Second, understanding CLF3 geometry provides insights into molecular bonding principles, which are fundamental to chemistry. Finally, this knowledge can be applied in industries where CLF3 is used, such as semiconductor manufacturing and aerospace.
Molecular Structure of CLF3
The molecular structure of CLF3 is determined by the arrangement of its constituent atoms. Chlorine, being the central atom, forms covalent bonds with three fluorine atoms. Additionally, chlorine has two lone pairs of electrons, which influence the overall geometry of the molecule.
- Central atom: Chlorine
- Bonding atoms: Three fluorine atoms
- Non-bonding electrons: Two lone pairs on chlorine
Hybridization in CLF3
What is Hybridization?
Hybridization refers to the mixing of atomic orbitals to form new hybrid orbitals, which are used in bonding. In the case of CLF3, the central chlorine atom undergoes sp3d hybridization. This hybridization results in five hybrid orbitals, four of which are involved in bonding with fluorine atoms, while one accommodates the lone pair.
Impact of Hybridization on Geometry
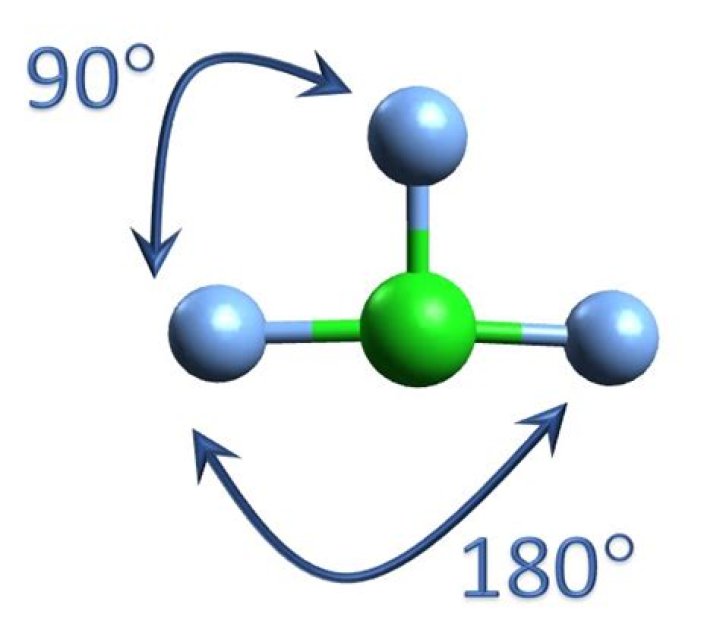
The sp3d hybridization in CLF3 leads to a trigonal bipyramidal electron geometry. However, due to the presence of lone pairs, the actual molecular geometry is distorted, resulting in a T-shaped structure. This distortion is a direct consequence of lone pair-bond pair repulsion, as explained by the VSEPR (Valence Shell Electron Pair Repulsion) theory.
Bond Angles in CLF3
The bond angles in CLF3 are influenced by the repulsion between lone pairs and bonding pairs of electrons. In a perfect trigonal bipyramidal arrangement, the bond angles would be 90° and 120°. However, the presence of lone pairs causes deviations from these ideal angles:
- F-Cl-F bond angles (equatorial): Approximately 87.5°
- F-Cl-F bond angles (axial): Approximately 175°
Shape of CLF3 Molecule
The shape of the CLF3 molecule is T-shaped. This shape arises from the trigonal bipyramidal electron geometry distorted by lone pair repulsion. The three fluorine atoms occupy the equatorial positions, while the two lone pairs occupy the axial positions. This arrangement minimizes electron repulsion, stabilizing the molecule.
Physical and Chemical Properties
CLF3 exhibits several notable physical and chemical properties:
- Appearance: Colorless gas
- Boiling Point: -8.9°C
- Melting Point: -76.3°C
- Density: 3.75 g/L (at 0°C)
- Reactivity: Highly reactive, acts as a strong oxidizing agent
These properties make CLF3 suitable for applications where strong oxidizing power is required.
Applications of CLF3
Industrial Uses
CLF3 is widely used in various industrial applications due to its reactivity:
- Etching agent in semiconductor manufacturing
- Propellant in rocket fuels
- Oxidizing agent in chemical reactions
Environmental Considerations
Despite its utility, CLF3 poses environmental risks due to its reactivity and potential to form harmful byproducts. Proper handling and disposal are critical to minimizing its impact on the environment.
Variations of CLF3 Geometry
While CLF3 exhibits a T-shaped geometry, variations in molecular geometry can occur due to external factors such as temperature, pressure, or the presence of other molecules. These variations can influence the compound's reactivity and stability. Understanding these variations is essential for optimizing its use in various applications.
Comparison with Other Molecules
Similarities with Other Halides
CLF3 shares similarities with other halides, such as bromine trifluoride (BrF3). Both compounds exhibit T-shaped geometries due to the presence of lone pairs on the central atom. However, differences in atomic size and electronegativity result in variations in bond angles and reactivity.
Differences from Non-Halides
Compared to non-halide molecules, CLF3's geometry is more complex due to the influence of lone pairs. Non-halide molecules, such as methane (CH4), exhibit simpler geometries due to the absence of lone pairs. This difference highlights the importance of lone pairs in determining molecular geometry.
Conclusion and Future Directions
In conclusion, CLF3 geometry is a critical aspect of understanding this highly reactive compound. Its T-shaped structure, resulting from sp3d hybridization and lone pair repulsion, influences its reactivity and applications. By studying CLF3 geometry, we gain valuable insights into molecular bonding principles and their practical implications.
We encourage readers to explore further topics in molecular geometry and chemistry. Share your thoughts in the comments section below, and don't forget to check out other articles on our website for more in-depth knowledge. Together, we can deepen our understanding of the fascinating world of chemistry.
Data sources:
- Chemistry textbooks and peer-reviewed journals
- Reputable online chemistry databases
- Industrial manuals and technical reports


