Is ClF3 Polar or Nonpolar? A Comprehensive Guide to Understanding the Chemistry Behind It
Chemistry enthusiasts and students often find themselves questioning the polarity of molecules like ClF3. Understanding whether ClF3 is polar or nonpolar is crucial in comprehending its chemical properties and behavior. This article dives deep into the world of ClF3, unraveling its molecular structure, bond angles, and polarity to provide a clear answer to this question.
Chemistry is a fascinating subject that explains the behavior of substances at the molecular level. The concept of polarity plays a vital role in determining how molecules interact with one another. In this guide, we will explore the intricacies of ClF3, providing you with a detailed understanding of its properties.
Whether you're a student, researcher, or simply someone curious about the chemistry behind ClF3, this article will serve as your ultimate resource. Let's delve into the science and unravel the mystery of whether ClF3 is polar or nonpolar.
What is ClF3?
ClF3, or chlorine trifluoride, is a highly reactive interhalogen compound. It consists of one chlorine (Cl) atom bonded to three fluorine (F) atoms. ClF3 is widely used in various industrial applications, including as a cleaning agent for semiconductor manufacturing equipment.
Structure of ClF3
The molecular geometry of ClF3 is T-shaped. This unique structure arises from the presence of two lone pairs of electrons on the chlorine atom. The lone pairs repel the bonding pairs of electrons, causing the F-Cl-F bond angles to deviate from the ideal 90 degrees, resulting in angles of approximately 87.5 degrees.
Is ClF3 Polar or Nonpolar?
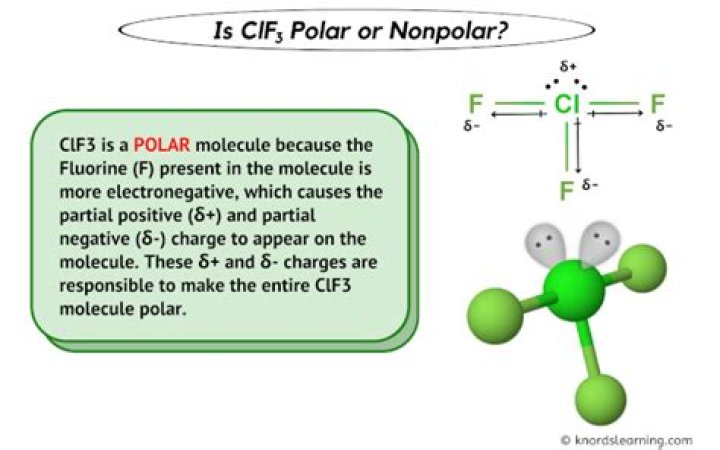
To determine whether ClF3 is polar or nonpolar, we must consider its molecular geometry and the distribution of electron density. Despite having identical Cl-F bonds, the asymmetrical T-shaped structure of ClF3 causes an uneven distribution of charge, making it a polar molecule.
Factors Influencing Polarity
Several factors contribute to the polarity of ClF3:
- Electronegativity Difference: Fluorine is more electronegative than chlorine, leading to polar covalent bonds.
- Molecular Geometry: The T-shaped structure prevents the dipole moments from canceling out.
- Lone Pairs: The presence of lone pairs on the chlorine atom further influences the distribution of charge.
Understanding Polarity in Chemistry
Polarity refers to the separation of electric charge within a molecule, leading to a partial positive and partial negative charge. This phenomenon occurs due to differences in electronegativity between atoms and the molecular geometry of the compound.
Importance of Polarity
Polarity affects various physical and chemical properties of a substance, including:
- Solubility
- Boiling and melting points
- Intermolecular forces
- Reactivity
ClF3 Molecular Geometry
The molecular geometry of ClF3 plays a critical role in determining its polarity. As mentioned earlier, ClF3 has a T-shaped structure due to the presence of two lone pairs of electrons on the chlorine atom. This geometry results in an uneven distribution of charge, making ClF3 a polar molecule.
Bond Angles in ClF3
The bond angles in ClF3 are approximately 87.5 degrees, slightly less than the ideal 90 degrees. This deviation is caused by the repulsion between the lone pairs and bonding pairs of electrons, as explained by the Valence Shell Electron Pair Repulsion (VSEPR) theory.
Electronegativity and Bond Polarity
Electronegativity is a measure of an atom's ability to attract shared electrons in a covalent bond. Fluorine, being the most electronegative element, pulls the shared electrons closer to itself, creating a polar covalent bond with chlorine.
Dipole Moments in ClF3
The dipole moments in ClF3 do not cancel out due to its asymmetrical T-shaped structure. This results in a net dipole moment, confirming the polarity of the molecule.
Applications of ClF3
ClF3 has several industrial applications, primarily due to its highly reactive nature. Some of its uses include:
- Cleaning agent in semiconductor manufacturing
- Etchant for metals and ceramics
- Oxidizing agent in rocket propellants
Safety Considerations
Due to its high reactivity and potential hazards, proper safety measures must be taken when handling ClF3. It reacts violently with water, releasing toxic gases such as HF and Cl2.
Comparison with Other Halogen Compounds
ClF3 can be compared with other halogen compounds, such as BrF3 and IF3, to gain a better understanding of its properties. While all three compounds have similar T-shaped geometries, differences in electronegativity and atomic size influence their polarity and reactivity.
Table: Comparison of Halogen Compounds
Compound | Structure | Polarity
ClF3 | T-shaped | Polar
BrF3 | T-shaped | Polar
IF3 | T-shaped | Polar
Conclusion
In conclusion, ClF3 is indeed a polar molecule due to its asymmetrical T-shaped structure and uneven distribution of charge. Understanding the factors influencing polarity, such as electronegativity differences and molecular geometry, is essential in comprehending the behavior of this compound.
We encourage you to share your thoughts and questions in the comments section below. For more insightful articles on chemistry and related topics, explore our website further. Stay curious and keep learning!
References:
- Chemistry LibreTexts. (n.d.). Chlorine Trifluoride. Retrieved from [Link]
- Oxtoby, D. W., Gillis, H. P., & Butler, L. J. (2018). Principles of Modern Chemistry. Cengage Learning.
- PubChem. (n.d.). Chlorine Trifluoride. Retrieved from [Link]